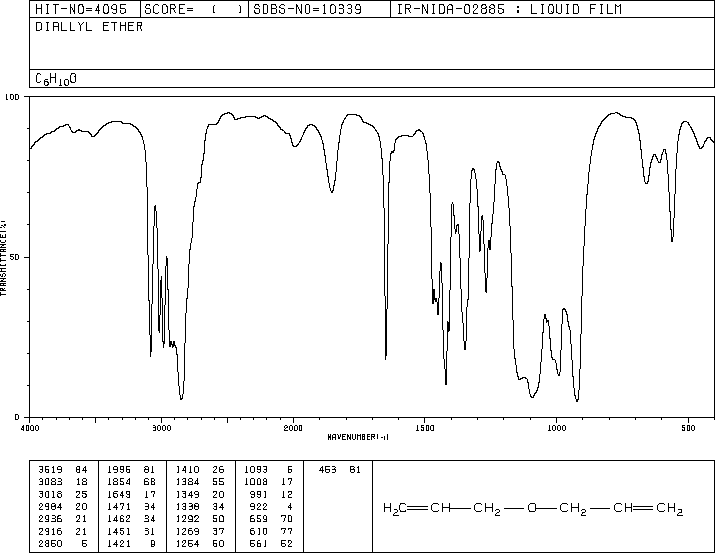
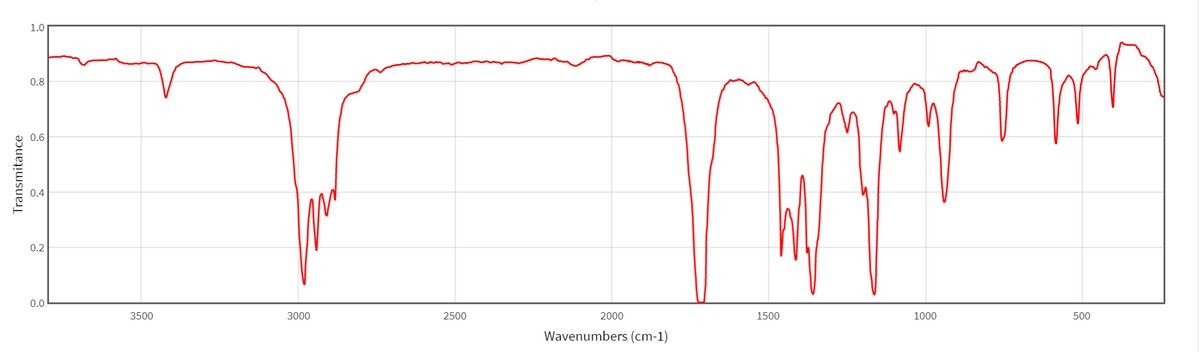
 Methoxy, methyl ether O-CH 3, C-H stretch 28502815 Methylamino, N-CH 3, C-H stretch 28202780 Figure 1. You couldn't be sure that this trough wasn't caused by something else. The schematic IR spectrum is available in Figure 1, and the specific frequency of each. The possible absorption due to the C-O single bond is queried because it lies in the fingerprint region. The infrared spectrum for ethanoic acid looks like this: It is easily recognised in an acid because it produces a very broad trough in the range 2500 - 3300 cm -1. This absorbs differently depending on its environment. The other really useful bond is the O-H bond.Its position varies slightly depending on what sort of compound it is in. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
